Project No.2261
Primary Supervisor
Dr David A Tumbarello – University of Southampton
Co-Supervisor(s)
Dr Lisa Mullen,- University of Sussex
Summary
Defects in mitochondrial quality control (mitoQC) are one of the primary drivers of cell death contributing to ageing and conditions such as Parkinson’s and cardiac disease.
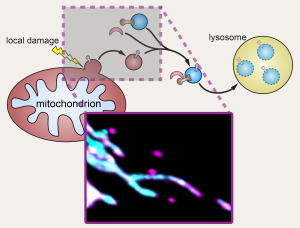
There is substantial literature linking inflammation and alterations in the innate immune response to mitoQC dysfunction and disease pathology. Recent evidence suggests defects in essential proteins that regulate mitoQC, such as PINK1 and Parkin, modulate the inflammatory response and exacerbate disease phenotypes. Our lab has identified Toll-interacting protein (Tollip) as a coordinator of a Parkin-
dependent mitochondrial derived vesicle (MDV) pathway that ensures damaged mitochondrial cargo is transported to the lysosome. Tollip was originally determined to be a negative regulator of Toll-like receptor signalling downstream of IL-1β, however more recently it has been identified as an organiser of endosomal positioning. Tollip’s essential role in MDV transport and its established function as a modulator of inflammatory signaling suggest it may represent a key link between these intimately connected pathways. This is additionally supported by recent work indicating Tollip directly regulates STING-mediated inflammation, which is a critical immune pathway downstream of the mitochondrial stress response. Therefore, this studentship aims to determine whether Tollip, in collaboration with Parkin, directly modulates innate immune regulators and inflammatory signaling in response to mitochondrial stress. The project will use cell models coupled to cellular imaging and biochemical methods to elucidate mechanisms that link mitochondrial stress to the innate immune response. The project will employ a variety of techniques including CRISPR-Cas9 editing approaches, in situ proximity dependent labelling to elucidate protein-protein interactions, microscopy, and biochemical assays to evaluate changes in inflammatory signalling. The outcomes will provide significant insight into new targets linking mitoQC to inflammation, influencing therapeutic development to treat disease. This project will also deliver important insight into how oxidative stress over a cell’s lifetime impact on multiple essential homeostatic pathways.